For example, the Li and Na atoms (in the first column) bond to other atoms in a similar way. Atoms that belong to the same column or chemical group share many of the same chemical properties. The table is read left to right and top to bottom in the order of increasing atomic number Z. (Recall that the total number of protons in an atomic nucleus is called the atomic number, Z.)įirst, the periodic table is arranged into columns and rows. In this discussion, we assume that the atoms are electrically neutral that is, they have the same number of electrons and protons. A detailed discussion of the periodic table is left to a chemistry course-we sketch only its basic features here. The structure of the periodic table ( Figure 8.17) can be understood in terms of shells and subshells, and, ultimately, the total energy, orbital angular momentum, and spin of the electrons in the atom. Before the n = 3 n = 3 shell is completely filled, for example, we begin to find electrons in the n = 4 n = 4 shell. 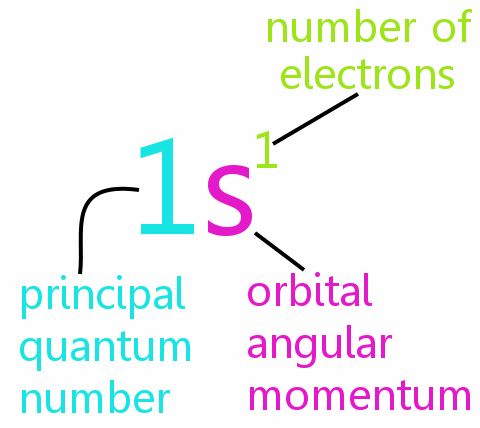
In standard (spectroscopic) notation, a filled n = 3 n = 3 shell is denoted as 3 s 2 3 p 6 3 d 10 3 s 2 3 p 6 3 d 10. The total number of electrons in the three possible subshells is thus the same as the formula 2 n 2 2 n 2. Maximum number of electrons = 2 n 2 = 2 ( 3 ) 2 = 2 ( 9 ) = 18. Chemical bonding between atoms in a molecule are explained by the transfer and sharing of valence electrons. Electrons in the outer shell of an atom are called valence electron s. The electron configurations for several atoms are given in Table 8.6. This representation of the electron state is called the electron configuration of the atom. The combination of two electrons in the n = 2 n = 2 and l = 0 l = 0 state, and three electrons in the n = 2 n = 2 and l = 1 l = 1 state is written as 2 s 2 2 p 3, 2 s 2 2 p 3, and so on. An electron in the n = 2 n = 2 state with l = 1 l = 1 is denoted 2 p. Two electrons in the n = 1 n = 1 state are denoted as 1 s 2, 1 s 2, where the superscript indicates the number of electrons. An electron in the n = 1 n = 1 state of a hydrogen atom is denoted 1 s, where the first digit indicates the shell ( n = 1 ) ( n = 1 ) and the letter indicates the subshell ( s, p, d, f … correspond to l = 0, 1, 2, 3 … ). Table 8.5 Electron States of Atoms Because of Pauli’s exclusion principle, no two electrons in an atom have the same set of four quantum numbers.Įlectrons with the same principal quantum number n are said to be in the same shell, and those that have the same value of l are said to occupy the same subshell. Consistent with Pauli’s exclusion principle, no two rows of the table have the exact same set of quantum numbers. Sample sets of quantum numbers for the electrons in an atom are given in Table 8.5. 
This principle is related to two properties of electrons: All electrons are identical (“when you’ve seen one electron, you’ve seen them all”) and they have half-integral spin ( s = 1 / 2 ). The structure and chemical properties of atoms are explained in part by Pauli’s exclusion principle: No two electrons in an atom can have the same values for all four quantum numbers ( n, l, m, m s ). (The spin quantum number s is the same for all electrons, so it will not be used in this section.) The electric potential U( r) for each electron does not follow the simple −1 / r −1 / r form because of interactions between electrons, but it turns out that we can still label each individual electron state by quantum numbers, ( n, l, m, s, m s ) ( n, l, m, s, m s ). The assumption is valid because the electrons are distributed randomly around the nucleus and produce an average electric field (and potential) that is spherically symmetrical. Assume that each electron moves in a spherically symmetrical electric field produced by the nucleus and all other electrons of the atom. To construct the ground state of a neutral multi-electron atom, imagine starting with a nucleus of charge Ze (that is, a nucleus of atomic number Z) and then adding Z electrons one by one. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
